Steel Production
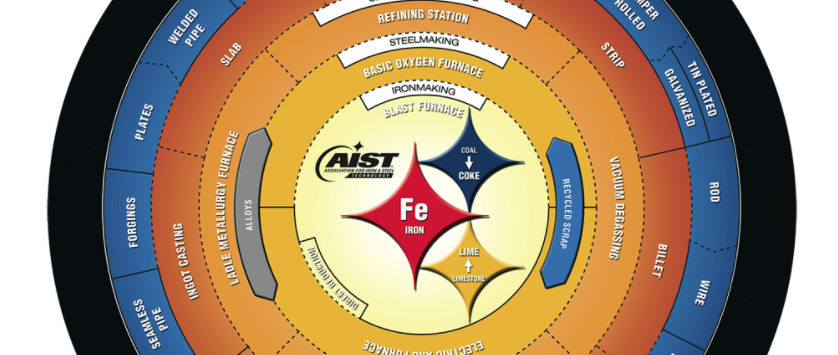
Steel Flowlines
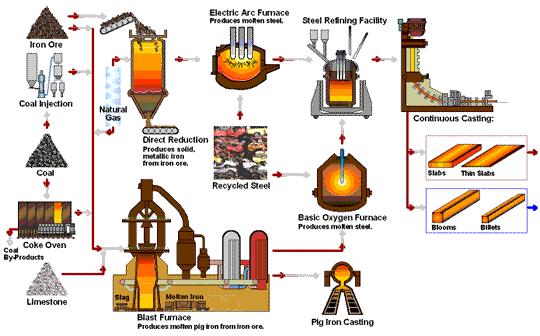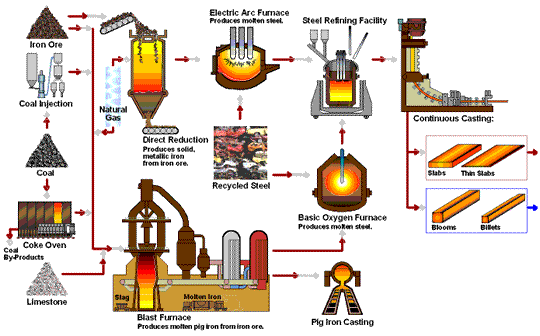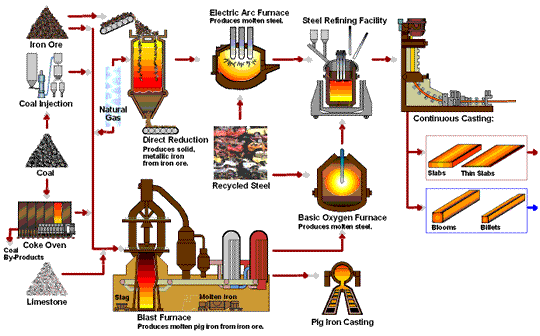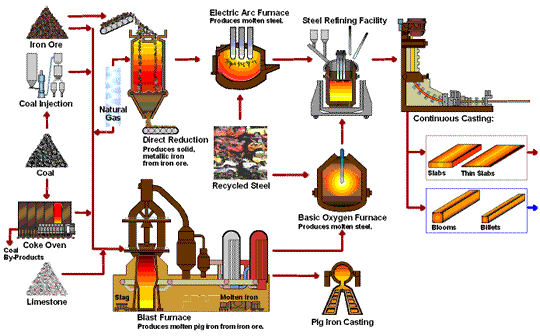
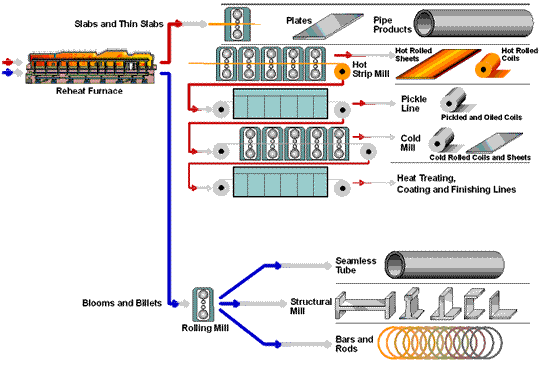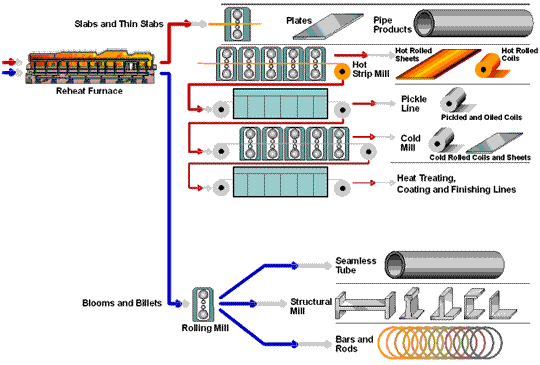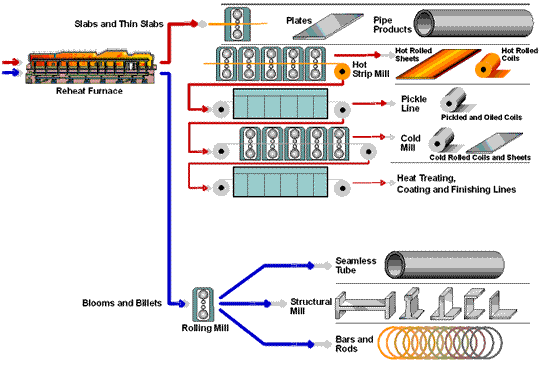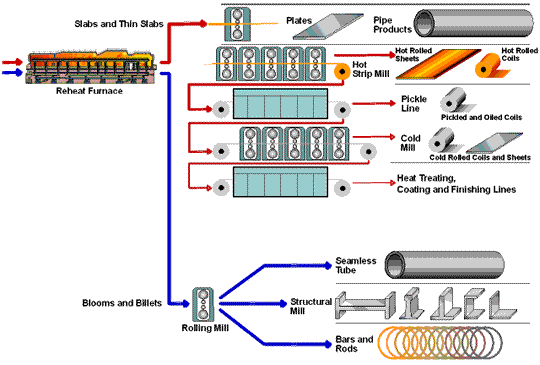
How Steel Is Made
 Steel is primarily produced using one of two methods: Blast Furnace or Electric Arc Furnace.
Steel is primarily produced using one of two methods: Blast Furnace or Electric Arc Furnace.
The blast furnace is the first step in producing steel from iron oxides. The first blast furnaces appeared in the 14th century and produced one ton per day. Even though equipment is improved and higher production rates can be achieved, the processes inside the blast furnace remain the same. The blast furnace uses coke, iron ore and limestone to produce pig iron.
Coal traditionally has been a key part of the coke-making process. The coal is crushed and ground into a powder and then charged into an oven where it is heated to approximately 1800°F in the absence of oxygen. As the oven is heated, the coal begins to melt so most of the volatile matter such as oil, tar, hydrogen, nitrogen and sulfur are removed. The cooked coal, called coke, is removed from the oven after 18 to 24 hours of reaction time. The coke is cooled and screened into pieces ranging from one inch to four inches. The coke is a porous, hard black rock of concentrated carbon (contains 90 to 93 percent carbon), which has some ash and sulfur but compared to raw coal is very strong. The strong pieces of coke with a high energy value provide permeability, heat and gases which are required to reduce and melt the iron ore, pellets and sinter. Today, natural gas is increasingly being added in place of coke to the same degree in the blast furnace to reduce carbon emissions.
The first electric arc furnaces (EAFs) appeared in the late 19th Century. The use of EAFs has expanded and now accounts for over 70 percent of steel production in the United States. The EAF is different from the blast furnace as it produces steel by using an electrical current to melt scrap steel, direct reduced iron, and/or pig iron, to produce molten steel.
![[AISI LOGO]](https://www.steel.org/wp-content/themes/steel-org/assets/images/steel-logo.png) American
Iron and Steel
Institute
American
Iron and Steel
Institute